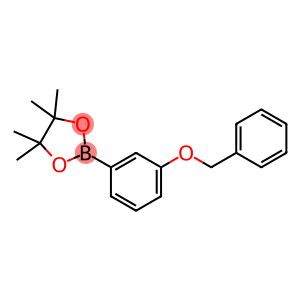
3-BENZYLOXYPHENYLBORONIC ACID, PINACOL ESTER
2-[3-(Benzyloxy)phenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane
CAS: 765908-38-1
Molecular Formula: C19H23BO3
3-BENZYLOXYPHENYLBORONIC ACID, PINACOL ESTER - Names and Identifiers
3-BENZYLOXYPHENYLBORONIC ACID, PINACOL ESTER - Physico-chemical Properties
| Molecular Formula | C19H23BO3 |
| Molar Mass | 310.2 |
| Density | 1.08±0.1 g/cm3(Predicted) |
| Melting Point | 58-62°C(lit.) |
| Boling Point | 434.9±28.0 °C(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Sensitive | Irritant |
| MDL | MFCD05663839 |
3-BENZYLOXYPHENYLBORONIC ACID, PINACOL ESTER - Risk and Safety
| WGK Germany | 3 |
3-BENZYLOXYPHENYLBORONIC ACID, PINACOL ESTER - Introduction
2-[3-(Benzyloxy)phenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane, often abbreviated as BOB or BOB entity, is an organoboron compound. Here are details about the compound:
Nature:
BOB is a solid with a colorless to pale yellow appearance. It is insoluble in water at room temperature, but soluble in many organic solvents, such as ether, methanol and methylene chloride. It is a stable compound that can be stored for a long time under normal pressure.
Use:
BOB is an important reagent in the field of organic synthesis. It is often used as a reagent for cross-coupling reactions and carbon-boron bond formation. BOB can be used as a substrate and reagent to participate in the arylation reaction of phenyl and the borylation reaction of thiol. In addition, BOB can also be used in the synthesis of organic hybrid materials and fluorescent dyes and other compounds.
Preparation Method:
The synthesis of BOB is usually obtained by a coupling reaction of a boron compound. A common synthetic method involves the reaction of p-tert-butoxyphenylboronate with benzyl alcohol, and the synthesis of aromatic hydrocarbon borates under base catalysis. Ethanol or dichloromethane is commonly used as a solvent for this reaction.
Safety Information:
BOB can be regarded as a relatively safe compound under the correct operation and storage, but the following matters still need to be paid attention. BOB is irritating to the eyes and skin, so it is necessary to use personal protective equipment such as protective gloves and goggles when in contact. When used in the laboratory, strict safety procedures should be followed and good ventilation should be ensured. At the same time, BOB should avoid contact with strong oxidants and strong acids. Because of its specific toxicity and other detailed safety information, it is still necessary to refer to the safety instructions and instructions for use of chemicals to ensure safe use. It is recommended that you do not use this compound on your own without training or certification.
Nature:
BOB is a solid with a colorless to pale yellow appearance. It is insoluble in water at room temperature, but soluble in many organic solvents, such as ether, methanol and methylene chloride. It is a stable compound that can be stored for a long time under normal pressure.
Use:
BOB is an important reagent in the field of organic synthesis. It is often used as a reagent for cross-coupling reactions and carbon-boron bond formation. BOB can be used as a substrate and reagent to participate in the arylation reaction of phenyl and the borylation reaction of thiol. In addition, BOB can also be used in the synthesis of organic hybrid materials and fluorescent dyes and other compounds.
Preparation Method:
The synthesis of BOB is usually obtained by a coupling reaction of a boron compound. A common synthetic method involves the reaction of p-tert-butoxyphenylboronate with benzyl alcohol, and the synthesis of aromatic hydrocarbon borates under base catalysis. Ethanol or dichloromethane is commonly used as a solvent for this reaction.
Safety Information:
BOB can be regarded as a relatively safe compound under the correct operation and storage, but the following matters still need to be paid attention. BOB is irritating to the eyes and skin, so it is necessary to use personal protective equipment such as protective gloves and goggles when in contact. When used in the laboratory, strict safety procedures should be followed and good ventilation should be ensured. At the same time, BOB should avoid contact with strong oxidants and strong acids. Because of its specific toxicity and other detailed safety information, it is still necessary to refer to the safety instructions and instructions for use of chemicals to ensure safe use. It is recommended that you do not use this compound on your own without training or certification.
Last Update:2024-04-09 21:11:58
Supplier List
Spot supply
Product Name: 2-(3-(Benzyloxy)phenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane Visit Supplier Webpage Request for quotationCAS: 765908-38-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-(3-BENZYLOXYPHENYL)-4,4,5,5-TETRAMETHYL-1,3,2-DIOXABOROLANE Request for quotation
CAS: 765908-38-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 765908-38-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 2-(3-(Benzyloxy)phenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane Visit Supplier Webpage Request for quotationCAS: 765908-38-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-(3-BENZYLOXYPHENYL)-4,4,5,5-TETRAMETHYL-1,3,2-DIOXABOROLANE Request for quotation
CAS: 765908-38-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 765908-38-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
View History

